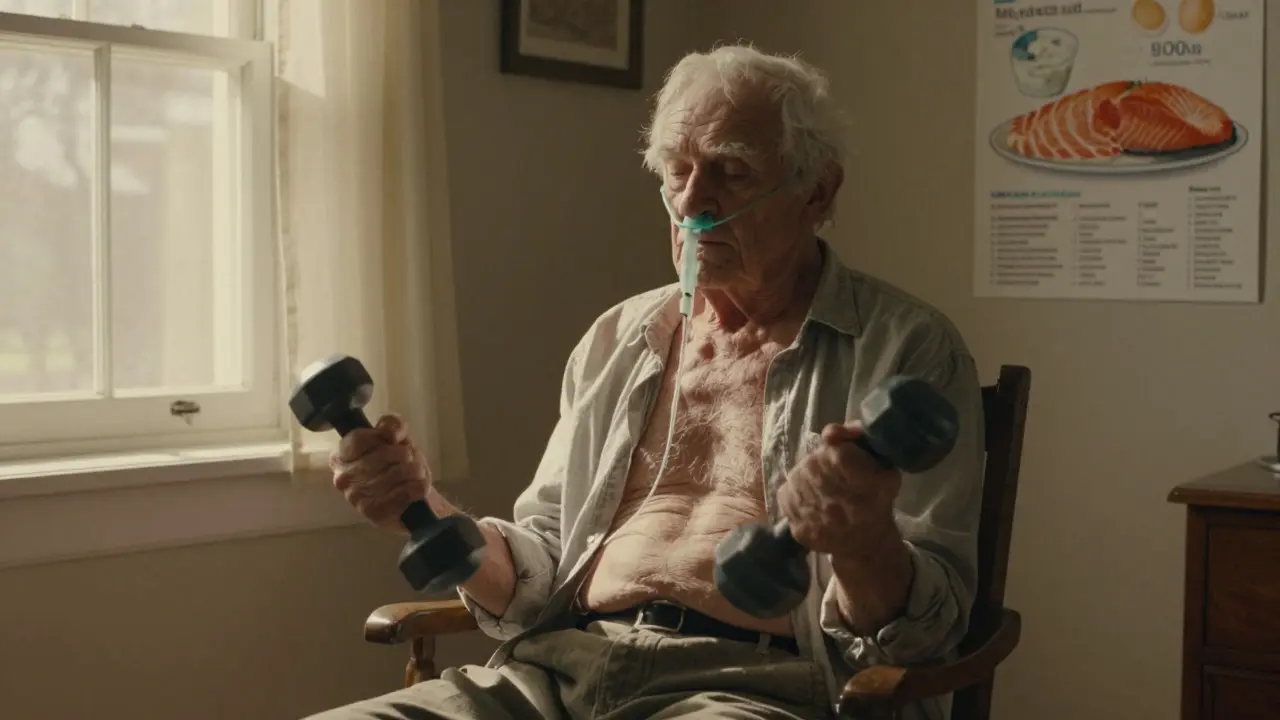Order-RxPills.com: Your Trusted Pharmaceutical Resource - Page 3
Fluoroquinolones and Tendon Rupture: What You Need to Know About the Risk
Fluoroquinolone antibiotics like ciprofloxacin and levofloxacin can cause sudden, serious tendon damage - especially in older adults and those on steroids. Learn who’s at risk, what to watch for, and how to avoid permanent injury.
Personnel Training: Qualifications for Manufacturing Staff
Manufacturing staff today need more than a high school diploma-they need certifications, digital skills, and safety training. Learn the key qualifications, certifications like CPT and Six Sigma, and how training is evolving to meet Industry 4.0 demands.
Telehealth Strategies for Monitoring Side Effects in Rural and Remote Patients
Telehealth is transforming care for rural patients by enabling real-time monitoring of medication side effects. With smart devices, AI alerts, and pharmacist-led programs, remote communities are seeing fewer hospitalizations and better medication adherence.
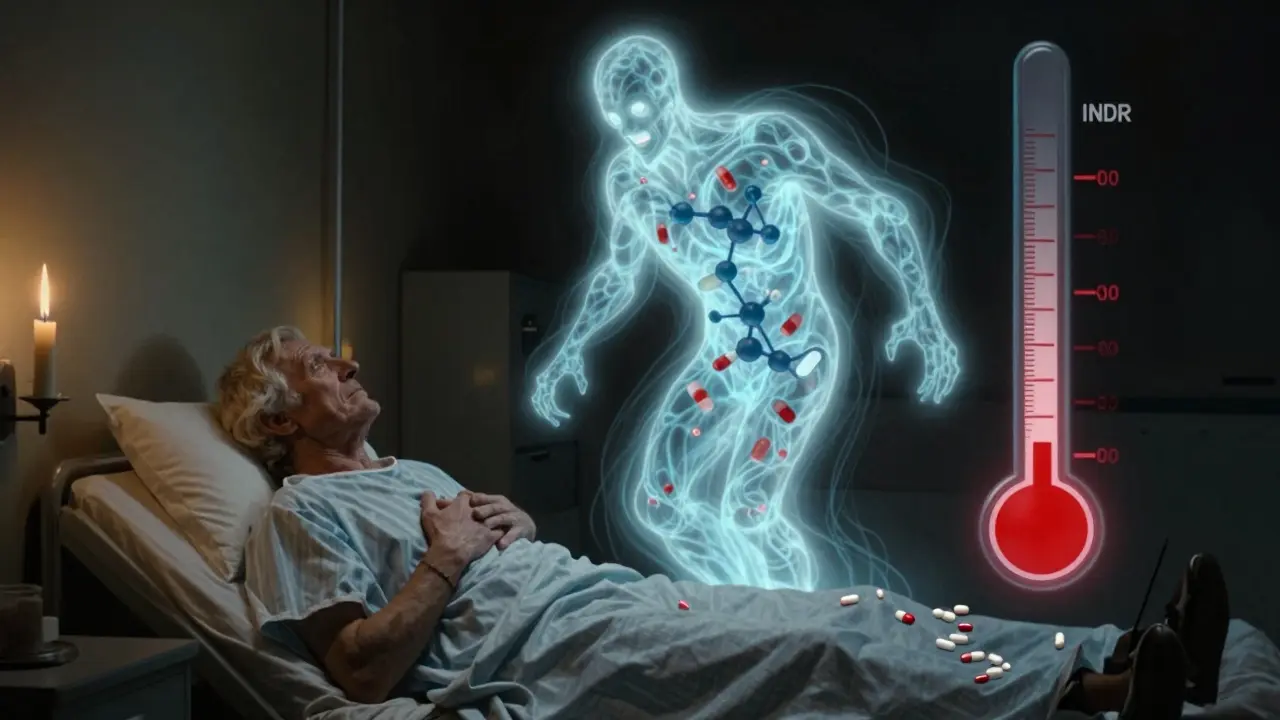
Trimethoprim-Sulfamethoxazole and Warfarin: How This Combo Raises INR and Risks Bleeding
Trimethoprim-sulfamethoxazole (Bactrim) can cause dangerous INR spikes in people taking warfarin, raising bleeding risk. Learn why this interaction happens, how to prevent it, and what to do if your INR rises.
Sarcopenia in COPD: How Nutrition and Resistance Training Can Improve Survival and Daily Function
Sarcopenia in COPD leads to faster muscle loss, higher hospitalization rates, and reduced survival. With proper protein intake and tailored resistance training, patients can rebuild strength, breathe easier, and live longer.
Medications Never to Put in Household Trash: A Safety List
Some medications are so dangerous they must be flushed down the toilet - not thrown in the trash. Learn which ones are on the FDA's safety list and how to dispose of them safely to prevent accidental overdoses.
How Clinician Communication Shapes Patient Beliefs About Generic Medications
Clinician communication plays a decisive role in whether patients trust and adhere to generic medications. Clear, confident explanations about FDA bioequivalence and nocebo effects can boost acceptance by over 30%, turning skepticism into confidence.
Patent Term Restoration Explained: How to Extend Patent Life for Drugs and Devices
Patent Term Restoration (PTE) helps drug developers recover lost patent time due to FDA approval delays. Learn how the Hatch-Waxman Act works, eligibility rules, current trends, and common pitfalls. With over 1,200 extensions granted between 2010-2020, PTE is crucial for pharmaceutical innovation.
Quality by Design in Generic Drug Development: Science-Based Approaches for Better Quality and Approval Rates
Quality by Design (QbD) revolutionizes generic drug development by integrating science and risk management from the start. Learn how it reduces FDA review times, cuts costs, and improves product quality. Real-world examples and future trends in this detailed guide.
MedWatch System Explained: How FDA Tracks Drug and Device Safety
MedWatch is the FDA's system for collecting safety reports on drugs, devices, and cosmetics after they reach the market. Learn how it works, who reports, and why your report matters.